Questions on Primate Behaviuor
- 1. Speculate on how the behavioral ecology of nonhuman primates may be helpful in explaining some human behaviors.
- 2. How might infanticide be seen as a reproductive strategy for males? What would you say if you saw a newspaper article that applied this idea (not the act itself) to human males? Do you think some people would object? Why or why not?
- 3. Do you think that knowing about aggression between groups of chimpanzees is useful in understanding conflicts
- between human societies? Why or why not?
- 4. Why are the language capabilities of nonhuman primates important to our understanding of how our own species may have acquired language?
The Evolution of Behavior
Scientists study behavior in free-ranging primates from an ecological and evolutionary perspective, focusing on the relationship between individual and social behaviors, the natural environment, and various physiological traits of the species in question. This approach is called behavioral ecology, and it’s based on the underlying assumption that all of the interconnected biological components of ecological systems (animals, plants, and microorganisms) evolved together. Therefore, behaviors are adaptations to environmental circumstances that existed in the past as well as in the present.
Briefly, the cornerstone of this perspective is that behaviors have evolved through the operation of natural selection and are therefore subject to natural selection in the same way physical traits are. (Remember that within a specific environmental context, natural selection favors characteristics that provide a reproductive advantage to the individuals who possess them.) Therefore, behavior constitutes a phenotype, and individuals whose behavioral phenotypes increase reproductive fitness will pass on their genes at a faster rate than others. But this doesn’t mean that primatologists think that genes code for specific behaviors, such as a gene for aggression, another for cooperation, and so on.
Studying complex behaviors from an evolutionary viewpoint doesn’t imply a one gene–one behavior relationship, nor does it suggest that behaviors that are influenced by genes can’t be modified through learning. In insects and other invertebrates, behavior is mostly under genetic control. In other words, most behavioral patterns in these species aren’t learned; they’re innate. But in many vertebrates, especially birds and mammals, the proportion of behavior that’s due to learning is substantially increased, while the proportion under genetic control is reduced. This is especially true of primates; and in humans, who are so much a product of culture, most behavior is learned. Still, we know that in mammals and birds, some behaviors are at least partly influenced by certain gene products, such as hormones. You may be aware of studies showing that increased levels of the hormone testosterone will increase aggression in many species. You may also know that abnormal levels of certain chemicals produced by brain cells can cause depression, schizophrenia, and bipolar disorder. Because brain cells are directed by the genes within them to produce these chemicals, this is another example of genes influencing behavior. Because behavioral genetics, or the study of how genes affect behavior, is a relatively new field, we don’t really know the extent to which genes actually influence behavior in humans or other species. But we do know that behavior must be viewed as the product of complex interactions between genetic and environmental factors. The limits and potentials for learning and for behavioral flexibility vary considerably among species. In some species, such as primates, the potentials are extremely broad; but in others, like insects, they aren’t. Ultimately, those limits and potentials are set by genetic factors that have been subjected to natural selection throughout the evolutionary history of every species. That history, in turn, has been shaped by the ecological setting not only of living species but also of their ancestors.
One of the major goals of primatology is to discover how certain behaviors influence reproductive fitness and how ecological factors have shaped the evolution of those behaviors. While the actual mechanics of behavioral evolution aren’t yet fully understood, new technologies and methodologies are helping scientists answer many questions. For example, genetic analysis has recently been used to establish paternity in a few primate groups, and this has helped support hypotheses about some behaviors in males. But in general, an evolution feeding or mating. To understand the function of one behavioral element, it’s necessary to determine how it’s influenced by numerous interrelated factors. As an example, we’ll consider some of the more important variables that influence social structure. Bear in mind that social structure itself influences individual behavior, so in many cases, the distinctions between social and individual behaviors are blurred.
Some Factors That Influence Social Structure
- Body Size As a rule, larger animals require fewer calories per unit of weight than smaller animals because larger animals have less surface area relative to body mass than smaller animals. Since body heat is lost at the surface, larger animals can retain heat more efficiently, so they need less energy overall. It may seem strange, but two 10-pound monkeys require more food than one 22-pound monkey (Fleagle, 1999).
- Basal Metabolic Rate(BMR) : The BMR concerns metabolism, the rate at which the body uses energy to maintain all bodily functions while in a resting state. It’s closely correlated with body size, so in general, smaller animals have a higher BMR than larger ones . Consequently, smaller primates, like galagos and marmosets, require an energy-rich diet high in protein (insects),
fats (nuts and seeds), and carbohydrates (fruits and seeds). Some larger primates, which tend to have a lower BMR and reduced energy requirements relative to body size, can do well with less energyrich foods, such as leaves. - Diet : Since the nutritional requirements of animals are related to the previous two factors, all three have evolved together. Therefore, when primatologists study the relationships between diet and behavior, they consider the benefits in terms of energy (calories) derived from various food items against the costs (energy expended) of obtaining and digesting them. While small bodied primates focus on high-energy foods, larger ones don’t necessarily need to. For instance, gorillas eat leaves, pith from bamboo stems, and other types of vegetation, and they don’t need to use much energy searching for food because they’re frequently surrounded by it . Some monkeys, especially colobines (colobus and langur species), are primarily leaf eaters. Compared with many other monkeys, they’re fairly large bodied. They’ve also evolved elongated intestines and pouched stomachs that enable them, with the assistance of intestinal bacteria, to digest the tough fibers and cellulose in leaves. Moreover, in at least two langur species, there’s a duplicated gene that produces an enzyme that further helps with digestion. This gene duplication isn’t found in other primates that have been studied, so the duplication event probably occurred after colobines and cercopithecines last shared a common ancestor (Zhang et al., 2002). Since having a second copy of the gene was advantageous to colobine ancestors who were probably already eating some leaves, natural selection favored it to the point that it was established in the lineage. (The discovery of this gene duplication is another example of how new technologies help explain behavior— in this case, dietary differences.)
- Distribution of Resources: Various kinds of foods are distributed in different ways. Leaves can be abundant and dense and will therefore support large groups of animals. Insects, on the other hand, may be widely scattered, and the animals that rely on them usually feed alone or with only one or two others. Fruits, nuts, and berries in dispersed trees and shrubs occur in clumps. These are most efficiently exploited by smaller groups of animals, so large groups frequently break up into smaller subunits while feeding. Such subunits may consist of one-male–multifemale groups (some baboons) or matrilines (macaques) Species that feed on abundantly distributed resources may also live in onemale groups, and because food is plentiful, these one-male units are able to join with others to form large, stable communities (for example, howlers and some baboons). To the casual observer, these communities can appear to be multimale-multifemale groups . Some species that depend on foods distributed in small clumps are protective of resources, especially if their feeding area is small enough to be defended. Some live in small groups composed of a mated pair (siamangs) or a female with one or two males marmosets and tamarins). Naturally, dependent offspring are also included. Lastly, foods such as fruits, nuts, and berries are only seasonally available, and primates that rely on them must eat a wide variety of items. This is another factor that tends to favor smaller feeding groups
- Predation Primates, depending on their size, are vulnerable to many types of predators, including snakes, birds of prey, leopards, wild dogs, and other primates. Their response to predation depends on their body size, social structure, and type of predator. Typically, where predation pressure is high and body size is small, large communities are advantageous. These may be multimale multifemale groups or congregations of one-male groups.
- Relationships with Other, Nonpredatory Species: Many primate species associate with other primate and nonprimate species for various reasons, including predator avoidance. When they do share habitats with other species, they exploit somewhat different resources.
- Dispersal : Dispersal is another factor that influences social structure and relationships within groups. As is true of most mammals, members of one sex leave the group in which they were born (their natal group) about the time they become sexually mature. Male dispersal is the most common pattern (ring-tailed lemurs, vervets, and macaques, to name a few). But female dispersal is seen in some colobus species, hamadryas baboons, chimpanzees, and mountain gorillas. In species where the basic social structure is a mated pair, offspring of both sexes either leave or are driven away by their parents (gibbons and siamangs).
Dispersal may have more than one outcome. When females leave, they join another group. Males may do likewise, but in some species (for example, gorillas), they may live alone for a time, or they may temporarily join an all-male “bachelor” group until they’re able to establish a group of their own. But the common theme is that individuals who disperse usually find mates outside their natal group. This has led primatologists to conclude that the most valid explanations for dispersal are related to two major factors: reduced competition between males for mates and, more importantly, the decreased likelihood of close inbreeding. - Life Histories: Life history traits are characteristics or developmental stages that typify members of a given species and therefore influence potential reproductive rates. Examples of life history traits include length of gestation, length of time between pregnancies (interbirth interval), period of infant dependency and age at weaning, age at sexual maturity, and life expectancy. Life history traits have important consequences for many aspects of social life, and they can also be critical to species survival. Species that live only a few years mature rapidly, reproduce within a year or two after birth, and have short interbirth intervals. Thus, shorter life histories are advantageous to species that live in marginal or unpredictable habitats because reproduction can occur at a relatively rapid rate (Strier, 2003).
Conversely, longer-lived species, such as gorillas, are better suited to stable environmental conditions. The extended life spans of the great apes, characterized by later sexual maturation and long interbirth intervals of three to five years, means that most females will raise only three or four offspring to maturity. Today, this slow rate of reproduction increases the threat of extinction because the great apes are being hunted at a rate that far outpaces their replacement capacities. - Activity Patterns Most primates are diurnal, but galagos, lorises, aye-ayes, tarsiers, and New World owl monkeys are nocturnal. Nocturnal primates tend to forage for food alone or in groups of two or three and many hide to avoid predators.
- Human Activities : virtually all nonhuman primate populations are now impacted by human hunting and forest clearing. These activities severely disrupt and isolate groups, reduce numbers, reduce resource availability, and eventually can cause extinction.
Why Be Social?
Group living exposes animals to competition with other group members for resources, so why don’t primates live alone? After all, competition can lead to injury or even death, and it’s costly in terms of energy expenditure. One widely accepted answer to this question is that the costs of competition are offset by the benefits of predator defense. Multimale multifemale groups are advantageous in areas where predation pressure is high, particularly in mixed woodlands and on open savannas. Leopards are the most significant predator of terrestrial primates , but the chances of escaping a leopard attack are greater for animals that live in groups, where there are several pairs of eyes looking about. (There really is safety in numbers.) Savanna baboons have long been cited as an example of these principles. They live in semiarid grassland and broken woodland habitats throughout subSaharan Africa. To avoid nocturnal predators, savanna baboons sleep in trees, but they spend much of the day on the ground foraging for food. If a predator appears, baboons flee back into the trees, but if they’re some distance from safety, adult males (and sometimes females) may join forces to chase the intruder. The effectiveness of male baboons in this regard should not be underestimated; they’ve been known to kill domestic dogs and even to attack leopards and lions. There is probably no single answer to the question of why primates live in groups. More than likely, predator avoidance is a major factor but not the only one. Group living evolved as an adaptive response to a number of ecological variables, and it has served primates well for a very long time.
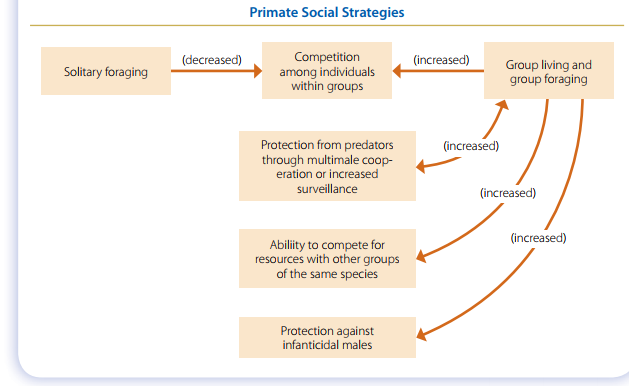
Primate Social Behavior
Because primates solve their major adaptive problems in a social context, we should expect them to behave in ways that reinforce the integrity of the group. The better known of these behaviors are described here. Remember, all these behaviors have evolved as adaptive responses during more than 50 million years of primate evolution.
Dominance
Many primate societies are organized into dominance hierarchies, which impose a certain degree of order by establishing parameters of individual behavior. Although aggression is frequently used to increase an animal’s status, dominance hierarchies usually serve to reduce the amount of actual physical violence. Not only are lower-ranking animals unlikely to attack or even threaten a higher-ranking one, but dominant animals are usually able to exert control simply by making a threatening gesture. Individual rank or status can be measured by access to resources, including food items and mating partners. Dominant animals (alpha males and females) are given priority by others, and they rarely give way in confrontations. Many primatologists think that the primary benefit of dominance is the increased reproductive success of high ranking animals. This is true in many cases, but there’s good evidence that lower-ranking males also successfully mate. High-ranking females also have higher reproductive success; because they have greater access to food than subordinate females, they’re able to obtain more energy for the production and care of offspring (Fedigan, 1983).
Pusey and colleagues (1997) demonstrated that the offspring of high ranking female chimpanzees at Gombe Stream National Park, in Tanzania, had significantly higher rates of infant survival. Moreover, their daughters matured faster, which meant they had shorter interbirth intervals and consequently produced more offspring.
An individual’s position in the hierarchy isn’t permanent and changes throughout life. It’s influenced by many factors, including sex, age, level of aggression, amount of time spent in the group, intelligence, perhaps motivation, and sometimes the mother’s social position (particularly true of macaques). In species organized into groups containing a number of females associated with one or several adult males, the males are generally dominant to females. Within such groups, males and females have separate hierarchies, although very high-ranking females can dominate the lowest-ranking males, particularly young ones. But there are exceptions to this pattern of male dominance. In many lemur species, females are the dominant sex. Moreover, in species that form bonded pairs (for example, indris and gibbons), males and females are codominant. All primates learn their position in the hierarchy. From birth, an infant is carried by its mother, and it observes how she responds to every member of the group. Just as important, it sees how others react to her. Dominance and subordination are indicated by gestures and behaviors, some of which are universal throughout the primate order, including humans, and this gestural repertoire is part of every youngster’s learning experience.
Young primates also acquire social rank through play with age peers, and as they spend more time with play groups, their social interactions widen. Competition and rough-and-tumble play allow them to learn the strengths and weaknesses of peers, and they carry this knowledge with them throughout their lives. Thus, through early contact with their mothers and subsequent exposure to peers, young primates learn to negotiate their way through the complex web of social interactions that make up their daily lives.


Communication
Communication is universal among animals and includes scents and unintentional, autonomic responses and behaviors that convey meaning. Such attributes as body posture provide information about an animal’s emotional state. For example, a purposeful striding gait implies confidence. Moreover, autonomic responses to threatening or novel stimuli, such as raised body hair (most species) or enhanced body odor (gorillas), indicate excitement. Many intentional behaviors also serve as communication. In primates, these include a wide variety of gestures, facial expressions, and vocalizations, some of which we humans share. Among many primates, an intense stare indicates a mild threat; and indeed, we humans find prolonged eye contact with strangers very uncomfortable. (For this reason, people should avoid eye contact with captive primates.) Other threat gestures are a quick yawn to expose canine teeth (baboons, macaques;); bobbing back and forth in a crouched position (patas monkeys); and branch shaking (many monkey species). High ranking baboons mount the hindquarters of subordinates to express dominance.
Mounting may also serve to defuse potentially tense situations by indicating something like, “It’s okay, I accept your apology.”
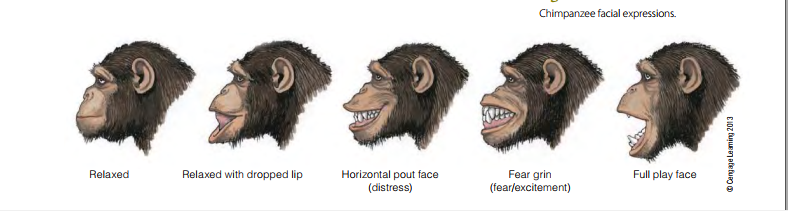
Primates also use a variety of behaviors to indicate submission, reassurance, or amicable intentions. Most primates crouch to show submission, and baboons also present or turn their hindquarters toward an animal they want to appease. Reassurance takes the form of touching, patting, hugging, and holding hands . Grooming also serves in a number of situations to indicate submission or reassurance. A wide variety of facial expressions indicating emotional state is seen in chimpanzees and, especially, in bonobos . These include the well-known play face (also seen in several other primate and nonprimate species), associated with play behavior, and the fear grin to indicate fear and submission. Not surprisingly, vocalizations play a major role in primate communication.
Some, such as the bark of a baboon that has just spotted a leopard, are unintentional startled reactions. Others, such as the chimpanzee food grunt, are heard only in specific contexts, in this case in the presence of food. These vocalizations, whether deliberate or not, inform others of the possible presence of predators or food. Primates (and other animals) also communicate through displays, which are more complicated, frequently elaborate combinations of behaviors. For example, the exaggerated courtship dances of many male birds, often enhanced by colorful plumage, are displays. Chest slapping and tearing vegetation are common gorilla threat displays.
All nonhuman animals use various body postures, vocalizations, and facial expressions to transmit information. But the array of communicative devices is much richer among nonhuman primates, even though they don’t use language the way humans do.
Communication is important because it’s what makes social living possible. Through submissive gestures, aggression is reduced and physical violence is less likely. Likewise, friendly intentions and relationships are reinforced through physical contact and grooming. Indeed, we humans can see ourselves in other primates most clearly in their use of nonverbal communication, particularly because some of their gestures and facial expressions carry the same meaning as ours do.
Aggressive Interactions
Within primate societies, there’s an interplay between aggressive behaviors, which can lead to group disruption, and affiliative behaviors, which promote group cohesion. Conflict within a group frequently develops out of competition for resources, including mating partners and food. Instead of actual attacks or fighting, most intragroup aggression occurs in the form of various signals and displays, frequently within the context of a dominance hierarchy. Therefore, the majority of tense situations are resolved through various submissive and appeasement behaviors. But conflicts aren’t always resolved peacefully; in fact, they can have serious and even fatal consequences. For example, high-ranking female macaques frequently intimidate, harass, and even attack lower-ranking females to keep them away from food. Dominant females consistently chase subordinates away from food and have even been observed taking food from their mouths. Eventually, these actions can cause weight loss and poor nutrition in low-ranking females and result in lower reproductive success; they’re less able to rear offspring to maturity because they simply don’t get enough to eat (Silk et al., 2003).
Competition between males for mates frequently results in injury and even death. In species that have a distinct breeding season, such as New World squirrel monkeys, conflict between males is most common during that time. In species not restricted to a mating season, such as baboons and chimpanzees, competition between males can be ongoing.
In recent years, some primatologists have focused on attacks by groups of animals on members of their own species. These conflicts occur when a number of individuals attack and sometimes kill one or two others who may or may not be members of the same group. Lethal aggression is relatively common between groups of chimpanzees, and it’s also been reported in several other species, including colobus monkeys, (Starin, 1994), spider monkeys (Aureli et al., 2006; Campbell, 2006), and capuchin monkeys (Gros-Louis et al., 2003). Between groups, aggression is often used to protect resources or territories. Primate groups are associated with a home range where they remain permanently. Although individuals may leave their home range and join another community, the group itself remains in a particular area. Within the home range is a portion called the core area, which contains the highest concentration of predictable resources, and it’s where the group is most frequently found. Although parts of a group’s home range may overlap with home ranges of other groups, core areas of adjacent groups don’t overlap. The core area can also be said to be a group’s territory, and it’s the portion of the home range defended against intrusion. However, in some species, other areas of the home range may also be defended . Not all primates are territorial. In general, territoriality is typical of species whose ranges are small enough to be patrolled and protected (gibbons and vervets, for example). But male chimpanzees are highly intolerant of unfamiliar chimpanzees, especially other males, and they fiercely defend their resources. Therefore, chimpanzee intergroup interactions are almost always characterized by aggressive displays, chasing, and sometimes very violent
fighting . Beginning in 1974, Jane Goodall and her colleagues witnessed at least five unprovoked and extremely brutal attacks by groups of chimpanzees on other chimpanzees (Goodall, 1986). To explain these attacks, it’s necessary to point out that by 1973, the original Gombe community had divided into two distinct groups, one located in the north and the other in the south of what had once been the original group’s home range. In effect, the smaller offshoot group had denied the others access to part of their former home range. By 1977, all seven males and one female of the splinter group were either known or suspected to have been killed. All observed incidents involved several animals, usually adult males, who brutally attacked lone individuals . Whether the chimpanzees actually intended to kill their victims is difficult to know, because we don’t know to what degree they have a concept of death. Mitani and colleagues (2010) document a similar situation in a large chimpanzee community at Ngogo, Kibale National Park, Uganda. Between 1999 and 2009, members of this group were observed killing or fatally wounding 18 individuals from other groups, and all but one of the observed attacks were made by coalitions of males on patrol. The entire Ngogo community now regularly uses the area where they frequently conducted border patrols and where 13 of the attacks occurred. Meanwhile, the former residents have not been seen. In effect, these chimpanzees have increased their territory by 2.5 square miles, or 22 percent. Given this fact, the researchers attribute the attacks on a neighboring group to territorial expansion, which increases their resource base and in turn may lead to greater reproductive success. Even though chimpanzees engage in lethal attacks, the actual number of observed incidents is low. The precise motivation of chimpanzee intergroup aggression may never be fully explained, but it appears that acquiring and protecting resources (including females) are involved (Nishida et al., 1985, 1990; Goodall, 1986; Manson and Wrangham, 1991; Nishida, 1991).
Through careful examination of shared aspects of human and chimpanzee social life, we can develop hypotheses regarding how intergroup conflict may have arisen in our own lineage. Early hominins and chimpanzees may have inherited from a common ancestor the predispositions that lead to similar patterns of strife between populations. It’s not possible to draw direct comparisons between chimpanzee conflict and modern human warfare owing to later human elaborations of culture: religion, use of symbols (such as flags), and language. But it’s important to speculate on the fundamental issues that may have led to the development of similar patterns in both species


Affiliation and Altruism
Even though conflict can be destructive, a certain amount of aggression helps protect resources and maintain order within groups. Fortunately, to minimize actual violence, promote group cohesion, and defuse potentially dangerous situations, there are many behaviors that reinforce bonds between individuals and enhance group stability. Common affiliative behaviors include reconciliation, consolation, and simple amicable interactions between friends and relatives. Most such behaviors involve various forms of physical contact, such as touching, handholding, hugging, and, among chimpanzees, kissing. In fact, physical contact is one of the most important factors in primate development, and it’s crucial in promoting peaceful relationships and reinforcing bonds in many primate social groups.
There are also behaviors that indicate just how important bonds between individuals are; and some of these behaviors can perhaps be said to be examples of caregiving, or compassion. It’s somewhat risky to use the term compassion because in humans, compassion is motivated by empathy for another person.
We don’t know for sure whether nonhuman primates can empathize with another’s suffering or misfortune, but laboratory research has indicated that some of them probably do. The degree to which chimpanzees and other primates are capable of empathy is debated by primatologists. Some believe that there is substantial evidence for it (deWaal, 1996, 2007), but others remain unconvinced (Silk et al., 2005). Certainly, there are many examples, mostly from chimpanzee studies, of caregiving actions that resemble compassionate behavior in humans.
Examples include protecting victims during attacks, helping younger siblings, and remaining near ill or dying relatives or friends. In a poignant example from Gombe, the young adult female Little Bee brought food to her mother at least twice while the latter lay dying of wounds inflicted by attacking males (Goodall, 1986). When chimpanzees have been observed sitting near a dying relative, they were seen occasionally to shoo flies away or groom the other, as if trying to help in some way.
Grooming is one of the most important affiliative behaviors in many primate species. Although grooming occurs in other animal species, social grooming is mostly a primate activity, and it plays an important role in day-to-day life . Because grooming involves using the fingers to pick through the fur of another individual (or one’s own) to remove insects, dirt, and other materials, it serves hygienic functions. But it’s also an immensely pleasurable activity that members of some species, especially chimpanzees, engage in for long periods of time. Grooming occurs in a variety of contexts. Mothers groom infants; males groom sexually receptive females; subordinate animals groom dominant ones, sometimes to gain favor; and friends groom friends. In general, grooming is comforting. It restores peaceful relationships after conflict and provides reassurance during tense situations. In short, grooming reinforces social bonds and consequently helps strengthen and maintain a group’s structure.
Conflict resolution through reconciliation is another important aspect of primate social behavior. Following a conflict, chimpanzee opponents frequently move, within minutes, to reconcile (de Waal, 1982). Reconciliation takes many forms, including hugging, kissing, and grooming. Even uninvolved individuals may take part, either grooming one or both participants or forming their own grooming parties. In addition, bonobos are unique in their use of sex to promote group cohesion, restore peace after conflicts, and relieve tension within the group (de Waal, 1987, 1989).
Social relationships are crucial to nonhuman primates, and bonds between individuals can last a lifetime. These relationships serve many functions. Individuals of many species form alliances in which members support each other against outsiders. Alliances, or coalitions, as they’re also called, can be used to enhance the status of members. For example, at Gombe, the male chimpanzee Figan achieved alpha status because of support from his brother (Goodall, 1986). In fact, chimpanzees so heavily rely on coalitions and are so skillful politically that an entire book, appropriately titled Chimpanzee Politics (de Waal, 1982), is devoted to the topic.
Altruism is behavior that benefits another while involving some risk or sacrifice to the performer. The most fundamental of altruistic behaviors, the protection of dependent offspring, is ubiquitous among mammals and birds, and in the majority of species, altruistic acts are confined to this context. Still, altruism, cooperation, and assistance are fairly common in many primate species, and altruistic acts sometimes contain elements of what might be interpreted as empathy. Chimpanzees routinely come to the aid of relatives and friends; female langurs join forces to protect infants from infanticidal males; and male baboons protect infants and cooperate to chase predators. In fact, the primate literature abounds with examples of altruistic acts, whereby individuals place themselves at some risk to protect others from attacks by conspecifics or predators.
Adopting orphans is a form of altruism that has been reported for capuchins, macaques, baboons, gorillas, and especially chimpanzees. When chimpanzee youngsters are orphaned, they are almost always adopted, usually by older siblings, who are attentive and highly protective. Adoption is crucial to the survival of orphans, who certainly wouldn’t survive on their own. In fact, it’s extremely rare for a chimpanzee orphan less than 3 years of age to survive, even if it is adopted. There are now hundreds of documented examples of cooperation and altruism in nonhuman primates. Chimpanzees certainly have shown a tendency to perform altruistic acts, and this fact has caused some primatologists to consider the possibility that the common ancestor of humans and chimpanzees had a propensity for cooperation and helping others, at least in certain circumstances (Warneken and Tomasello, 2006).
Evolutionary explanations of altruism are usually based on the premise that individuals are more likely to perform risky or self-sacrificing behaviors that benefit a relative. Therefore, by helping a relative who otherwise might not survive to reproduce, the performer is helping promote the spread of genes they have in common. Another explanation, sometimes called “reciprocal altruism,” emphasizes that performers help others to increase the chances that, at a future date, the recipient might return the favor.

Reproduction and Reproductive Behaviors
In most primate species, sexual behavior is tied to the female’s reproductive cycle, with females being receptive to males only when they’re in estrus. Estrus is characterized by behavioral changes that indicate that a female is receptive. In Old World monkeys and apes that live in multimale groups, estrus is also accompanied by swelling and changes in color of the skin around the genital area.
These changes serve as visual cues of a female’s readiness to mate . Permanent bonding between males and females isn’t common among nonhuman primates. However, male and female savanna baboons sometimes form mating consortships. These temporary relationships last while the female is in estrus, and the two spend most of their time together, mating frequently. Mating consortships are also sometimes seen in chimpanzees and are common in bonobos. In fact, male and female bonobos may spend several weeks primarily in each other’s company. During this time, they mate often, even when the female isn’t in estrus. However, these relationships of longer duration aren’t typical of chimpanzee (Pan troglodytes) males and females.
Such a male-female bond may result in increased reproductive success for both sexes. For the male, there is the increased likelihood that he will be the father of any infant the female conceives. At the same time, the female potentially gains protection from predators or other members of her group; and she may also gain some help in caring for offspring she may already have.
Female and Male Reproductive Strategies
Reproductive strategies, especially how they differ between the sexes, have been a primary focus of primate research. The goal of these strategies is to produce and successfully rear to adulthood as many offspring as possible. Primates are among the most K-selected of mammals. By this we mean that individuals produce only a few young, in whom they invest a tremendous amount of parental care. Contrast this pattern with r-selected species, where individuals produce large numbers of offspring but invest little or no energy in parental care. Good examples of r-selected species include insects, most fishes, and, among mammals, mice and rabbits.
Considering the degree of care required by young, dependent primate offspring, it’s clear that enormous investment by at least one parent is necessary, and in a majority of species, the mother carries most of the burden certainly before, but also after birth. Primates are totally helpless at birth, and because they develop slowly, they’re exposed to expanded learning opportunities within a social environment. This trend has been elaborated most dramatically in great apes and humans, especially the latter. So, what we see in ourselves and our close primate relatives (and presumably in our more recent ancestors as well) is a strategy in which at least one parent, usually the mother, makes an extraordinary investment to produce a few “high-quality,” slowly maturing offspring.
Finding food and mates, avoiding predators, and caring for and protecting dependent young are difficult challenges for nonhuman primates. Moreover, in most species, males and females use different strategies to meet these challenges. Female primates spend almost all their adult lives either pregnant, lactating, and/or caring for offspring, and the resulting metabolic demands are enormous. A pregnant or lactating female, although perhaps only half the size of her male counterpart, may require about the same number of calories per day.
Even if these demands are met, her physical resources may be drained. For example, analysis of chimpanzee skeletons from Gombe showed significant loss of bone and bone mineral in older females (Sumner et al., 1989).
Given these physiological costs and the fact that her reproductive potential is limited by lengthy intervals between births, a female’s best strategy is to maximize the amount of resources available to her and her offspring. Indeed, as we just discussed, females of many primate species are highly competitive with other females and aggressively protect resources. In other species, females distance themselves from others to avoid competition. Males, however, face a different set of challenges. Having little investment in the rearing of offspring along with the ability to produce sperm continuously, it’s to the male’s advantage to secure as many mates and produce as many offspring as possible.


Sexual Selection
Sexual selection is an outcome of different mating strategies. First described by Charles Darwin, it is a type of natural selection that operates on only one sex, usually males. The selective agent is male competition for mates and, in some species, mate choice by females. The long-term effect of sexual selection is to increase the frequency of those traits in males that lead to greater success in acquiring mates.
In the animal kingdom, numerous male attributes are the results of sexual selection. For example, female birds of many species are attracted to males with more vividly colored plumage. Selection has thus increased the frequency of alleles that influence brighter coloration in males, and in these species (peacocks are a good example), males are more colorful than females. Sexual selection in primates is most common in species in which mating is polygynous and there is considerable male competition for females. In these species, sexual selection produces dimorphism with regard to a number of traits, most noticeably body size . As you’ve seen, the males of many primate species are considerably
Infanticide as a Reproductive Strategy?
One way males may increase their chances of reproducing is to kill infants fathered by other males. This explanation was first offered in an early study of Hanuman langurs in India (Hrdy, 1977). Hanuman langurs typically live in groups composed of one adult male, several females, and their offspring. Other males without mates form “bachelor” groups that frequently forage within sight of one male–multifemale units. These peripheral males occasionally attack and defeat a reproductive male and drive him from his group. Sometimes, following such a takeover, the new male kills some or all of the group’s infants, fathered by the previous male.
At first glance, such behavior would seem to be counterproductive, especially for a species as a whole. However, individuals act to maximize their own reproductive success, no matter what effect their actions may have on the group or the species. By killing infants fathered by other animals, male langurs may in fact increase their own chances of fathering offspring, albeit unknowingly. This is because while a female is producing milk and nursing an infant, she doesn’t come into estrus and therefore isn’t sexually available. But when a female loses an infant, she resumes cycling and becomes sexually receptive. So, by killing nursing infants, a new male avoids waiting two to three years for them to be weaned before he can mate with their mothers. This could be advantageous for him because chances are good that he won’t even be in the group for two or three years. He also doesn’t expend energy and put himself at risk defending infants who don’t carry his genes. Hanuman langurs aren’t the only primates that practice infanticide.
Infanticide has been observed or surmised in many species, including gorillas, chimpanzees (Struhsaker and Leyland, 1987), and humans. In the majority of reported nonhuman primate examples, infanticide coincides with the transfer of a new male into a group or, as in chimpanzees, an encounter with an unfamiliar female and infant. (It should also be noted that infanticide occurs in numerous nonprimate species, including rodents, cats, and horses.) Numerous objections to this explanation of infanticide have been raised. Alternative explanations have included competition for resources (Rudran, 1973), aberrant behaviors related to human-induced overcrowding (Curtin and Dohlinow, 1978), and inadvertent killing during conflict between animals (Bartlett et al., 1993). Sussman and colleagues (1995), as well as others, have questioned the actual prevalence of infanticide, arguing that although it occurs, it’s not particularly common. These authors have also suggested that if indeed male reproductive fitness is increased through the killing of infants, such increases are negligible. Yet others (Struhsaker and Leyland, 1987; Hrdy et al., 1995) maintain that the incidence and patterning of infanticide by males are not only significant, but also consistent with the assumptions established by theories of behavioral evolution. More recently, Henzi and Barrett (2003, p. 224) reported that when chacma baboon males migrate into a new group, they “deliberately single out females with young infants and hunt them down” . The conclusion is that, at least in chacma baboons, newly arrived males consistently try to kill infants, and their attacks are highly aggressive. However, reports like these don’t prove that infanticide increases a male’s reproductive fitness. To do this, primatologists must demonstrate two crucial facts:
1. Infanticidal males don’t kill their own offspring.
2. Once a male has killed an infant, he subsequently fathers another infant with the victim’s mother.
These statements are hypotheses that can be tested; and to do this, Borries and colleagues (1999) collected DNA samples from the feces of infanticidal males and their victims’ remains in several groups of free-ranging Hanuman langurs. This was done to determine if these males killed their own offspring. The results showed that in all 16 cases where infant and male DNA was available, the males were not related to the infants they either attacked or killed. Moreover, DNA analysis also showed that in four out of five cases where a victim’s mother subsequently gave birth, the new infant was fathered by the infanticidal male.
The application of DNA technology to a long-unanswered question has provided strong evidence suggesting that infanticide may indeed give males an increased chance of fathering offspring. Moreover, this study provides another example of how hypotheses are further tested as new technologies are developed.


Mothers, Fathers, and Infants
The basic social unit among all primates is a female and her infants . Except in those species in which monogamy or polyandry occurs or the social group is a bonded pair, males usually don’t directly participate in the rearing of offspring. The mother-infant bond begins at birth. Although the exact nature of the bonding process isn’t fully understood, there appear to be predisposing innate factors that strongly attract the female to her infant, so long as she herself has had a sufficiently normal experience with her own mother. This doesn’t mean that primate mothers have innate knowledge of how to care for an infant. They don’t Monkeys and apes raised in captivity without contact with their own mothers not only don’t know how to care for a newborn infant, but may reject or even injure it. Thus, learning is essential to establishing a mother’s attraction to her infant. The role of bonding between primate mothers and infants was clearly demonstrated in a famous series of experiments at the University of Wisconsin.
Psychologist Harry Harlow (1959) raised infant rhesus macaques with surrogate mothers made of wire or a combination of wire and cloth. Other monkeys were raised with no mother at all. In one experiment, infants retained an attachment to their cloth-covered surrogate mother . But those raised with no mother were incapable of forming lasting attachments with other monkeys. None of the motherless males ever successfully copulated, and those females who were (somewhat artificially) impregnated either paid little attention to their infants or were aggressive toward them (Harlow and Harlow, 1961). The point is that monkeys reared in isolation were denied opportunities to learn the rules of social and maternal behavior. Moreover, and just as essential, they were denied the all-important physical contact so necessary for normal primate psychological and emotional development.
The importance of a normal relationship with the mother is demonstrated by field studies as well. From birth, infant primates are able to cling to their mother’s fur, and they’re in more or less constant physical contact with her for several months. During this critical period, infants develop closeness with their mothers that doesn’t always end with weaning. It may even be maintained throughout life. In some species, presumed fathers also participate in infant care . Male siamangs are actively involved, and marmoset and tamarin infants are usually carried on the father’s back and transferred to their mother only for nursing.


Primate Cultural Behavior
Cultural behavior is one important trait that makes primates, and especially chimpanzees and bonobos, attractive as models for behavior in early hominins. Although many cultural anthropologists and others prefer to apply the term culture specifically to human activities, most biological anthropologists consider it appropriate to apply the term to many nonhuman primate behaviors too (McGrew, 1992, 1998; de Waal, 1999; Whiten et al., 1999). Undeniably, most aspects of culture are uniquely human, and we should be cautious when we try to interpret nonhuman animal behavior. But again, since humans are products of the same evolutionary forces that have produced other species, they can be expected to exhibit some of the same behavioral patterns seen in other primates. However, because of increased brain size and learning capacities, humans express many characteristics to a greater degree, and culture is one of those characteristics. Cultural behavior is learned. In other words, it’s not genetically determined, although the capacity to learn is genetically influenced. Whereas humans deliberately teach their young, free-ranging nonhuman primates (with the exception of a few reports) don’t appear to do so. But at the same time, like young nonhuman primates, human children also acquire a tremendous amount of knowledge through observation rather than instruction . By watching their mothers and other members of their group, nonhuman primate infants learn about food items, appropriate behaviors, and how to use and modify objects to achieve certain ends. In turn, their own offspring will observe their activities.
What emerges is a cultural tradition that may eventually come to typify an entire group or even a species. The earliest reported example of cultural behavior concerned a study group of Japanese macaques on Koshima Island, Japan. In 1952, Japanese researchers began feeding the macaques sweet potatoes. The following year, a young female started washing her potatoes in a freshwater stream before eating them. Within three years, several monkeys were washing their potatoes, though instead of using the stream, they were taking their potatoes to the ocean nearby. Maybe they liked the salt.
The researchers pointed out that dietary habits and food preferences are learned and that potato washing is an example of nonhuman culture. Because the practice arose as an innovative solution to a problem (removing dirt) and gradually spread through the troop until it became a tradition, it was seen as containing elements of human culture. A study of orangutans listed 19 behaviors that showed sufficient regional variation to be classed as “very likely cultural variants” (van Schaik et al., 2003). Four of these were differences in how nests were used or built. Other behaviors that varied included the use of branches to swat insects and pressing leaves or hands to the mouth to amplify sounds.
Reports of tool use by gorillas aren’t common, but Breuer and colleagues (2005) reported seeing two female lowland gorillas in the DRC using branches as tools. In one case, a gorilla used a branch to test the depth of a pool of water. Then, as she waded bipedally through the pool, she used the branch again, this time as a walking stick . Chimpanzees exhibit more complex forms of tool use than any other nonhuman primate. This point is very important, because traditionally, tool use (along with language) was said to set humans apart from other animals. Chimpanzees crumple and chew handfuls of leaves, which they dip into tree hollows where water accumulates. Then they suck the water from the newly made “leaf sponges.” Leaves are also used to wipe substances from fur, and twigs are used as toothpicks, stones as weapons, and objects such as branches and stones may be dragged or rolled to enhance displays. “Termite fishing” is a common behavior among many chimpanzee groups. Chimpanzees routinely insert twigs and grass blades into termite mounds. The termites then seize the twig, and unfortunately for them, they become a light snack once the chimpanzee pulls the twig out of the mound. Chimpanzees also modify some of their stems by stripping the leaves or breaking them until they’re the right length. In effect, this is making a tool, and chimpanzees have Second, attention to the shape and size of the raw material indicates that chimpanzees have a preconceived idea of what the finished product needs to be in order to be useful. To produce a tool, even a simple one, based on a concept is an extremely complex behavior that, as we now know, is not the exclusive domain of humans.
Primatologists have been aware of termite fishing and similar behaviors since the 1960s, but they were surprised by the discovery that chimpanzees also use tools to catch small prey. Pruetz and Bertolani (2007) reported that savanna chimpanzees in Senegal, West Africa, sharpen small branches to use as thrusting spears for capturing galagos. This is the first report of a nonhuman primate hunting with what is basically a manufactured weapon. On 22 occasions, 10 different animals jabbed sharpened sticks into cavities in branches and trunks to extract galagos from their sleeping nests. In much the same way they modify termiting sticks, these chimpanzees had stripped off side twigs and leaves. But they’d also chewed the ends to sharpen them, in effect producing small thrusting spears. The spears weren’t necessarily used to impale victims so much as to injure or immobilize them because galagos are extremely agile and hard to catch. After several thrusts, the chimpanzee would reach into the opening to see if there was anything to be had. Observers only saw one galago being retrieved and eaten, and although it wasn’t moving or vocalizing, it was unclear if it had actually been killed by the “spear” (Pruetz and Bertolani, 2007). In several West African study groups, chimpanzees use unmodified stones as hammers and anvils to crack nuts and hard-shelled fruits (Boesch et al., 1994).
Stone hammers and platforms are used only in West African groups and not in East Africa. Likewise, termite fishing is seen in Central and East Africa, but apparently it’s not done in West African groups (McGrew, 1992). And using sharpened sticks to capture prey has been seen only in Senegal. The fact that chimpanzees show regional variation in the types of tools they use is significant because these differences represent cultural variation from one area to another. Chimpanzees also show regional dietary references (Nishida et al., 1983; McGrew, 1992, 1998). For example, oil palm fruits and nuts are eaten at many locations, including Gombe. But even though oil palms also grow in the Mahale Mountains (only about 90 miles from Gombe), the chimpanzees there seem to ignore them. Such regional patterns in tool use and food preferences are reminiscent of the cultural differences that are typical of humans. Therefore, it’s likely that this kind of variation probably existed in early hominins, too. So far, we’ve focused on tool use and culture in great apes, but they aren’t the only nonhuman primates that consistently use tools and exhibit elements of cultural behavior. Primatologists have been studying tool use in capuchin (also called cebus) monkeys for over 30 years. Capuchins are found in South America from Colombia and Venezuela, through Brazil, and as far south as northern Argentina. They have the largest relative brain size of all monkeys, and while forest-dwelling capuchin species are arboreal, other species live in a more savanna-like habitat and spend a fair amount of time on the ground. Many of the tool-using behaviors parallel those we’ve discussed for chimpanzees.
Capuchins use leaves to extract water from cavities in trees (Phillips, 1998), and they use small, modified branches to probe into holes in logs for invertebrates (Westergaard and Fragaszy, 1987). But what they’ve really become known for is using stones to obtain food. They use stones to smash foods into smaller pieces and crack palm nuts; break open hollow tree branches and logs; and dig for tubers and insects. Capuchins are the only monkeys known to use stones as tools and the only nonhuman primate to dig with stones (Visalberghi, 1990; Moura and Lee, 2004; Ottoni and Izar, 2008). The importance of palm nuts as a food source is revealed by the enormous effort expended to obtain it. Adult female and male capuchins weigh around 6 and 8 pounds, respectively, yet they walk bipedally carrying stones that weigh as much as 2 pounds, or 25 to 40 percent of their own body weight (Fragaszy et al., 2004; Visalberghi et al., 2007). Because the stones are heavy, it’s difficult for capuchins to sit while cracking nuts, so they frequently stand bipedally, raise the hammer stone with both hands, and then pound the nut using their entire body Even though chimpanzees and capuchins modify sticks to make tools, they haven’t been observed modifying the stones they use.
However, a male bonobo named Kanzi (see also p. 169) learned to strike two stones together to produce sharp-edged flakes. In a study con ducted by Sue Savage-Rumbaugh and archaeologist Nicholas Toth, Kanzi was allowed to watch as Toth produced stone flakes, which were then used to open a transparent plastic food container (Savage-Rumbaugh and Lewin, 1994). Bonobos don’t commonly use objects as tools in the wild. But Kanzi readily appreciated the usefulness of the flakes to get food. What’s more, he was able to master the basic technique of producing flakes without being taught, although at first his progress was slow. But then he realized that if he threw the stone onto a hard floor, it would shatter and he’d have lots of cutting tools. Although his solution wasn’t the one that Savage Rumbaugh and Toth had expected, it was even more significant because it provided an excellent example of bonobo insight and problem-solving ability. Kanzi did eventually learn to produce flakes by striking two stones together, and then he used these flakes to obtain food. These behaviors aren’t just examples of tool manufacture and use, albeit in a captive situation; they’re also very sophisticated goal-directed activities. Culture has become the environment in which modern humans live. Quite clearly, the use of sticks in termite fishing and hammer stones to crack nuts is hardly comparable to modern human technology. However, modern human technology had its beginnings in these very types of behaviors. But this doesn’t mean that nonhuman primates are “on their way” to becoming human. Remember, evolution isn’t goal directed, and even if it were, there’s nothing to dictate that modern humans necessarily constitute an evolutionary goal. Such a conclusion is a purely anthropocentric view and has no validity in discussions of evolutionary processes.




Language
One of the most significant events in human evolution was the development of language. We’ve already described several behaviors and autonomic responses that convey information in primates. But although we emphasized the importance of communication to nonhuman primate social life, we also said that nonhuman primates don’t use language the way humans do.
The view traditionally held by most linguists and behavioral psychologists was that nonhuman communication consists of mostly involuntary vocalizations and actions that convey information solely about the emotional state of the animal (anger, fear, and so on). Nonhuman animals haven’t been considered capable of communicating about external events, objects, or other animals, either in close proximity or removed in space or time. For example, when a startled baboon barks, other group members know only that it’s startled. But they don’t know what startled it, and they can only determine this by looking around to find the cause. In general, then, it’s been assumed that in nonhuman animals, including primates, vocalizations, facial expressions, body postures, and so on, don’t refer to specific external phenomena.
But for several years, these views have been challenged (Steklis, 1985; King, 1994, 2004). For example, vervet monkeys use specific vocalizations to refer to particular categories of predators, such as snakes, birds of prey, and leopards (Struhsaker, 1967; Seyfarth, Cheney, and Marler, 1980a, 1980b). When researchers made tape recordings of various vervet alarm calls and played them back within hearing distance of wild vervets, they saw different responses to various calls. When they heard leopard-alarm calls, the monkeys climbed trees; they looked up when they heard eagle-alarm calls; and they responded to snake-alarm calls by looking around at the ground.
These results show that vervets use distinct vocalizations to refer to specific components of the external environment. These calls aren’t involuntary, and they don’t refer solely to the emotional state (alarm) of the individual, although this information is conveyed. While these findings dispel certain long-held misconceptions about nonhuman communication (at least for some species), they also indicate certain limitations. Vervet communication is restricted to the present; as far as we know, no vervet can communicate about a predator it saw yesterday or one it might see tomorrow.
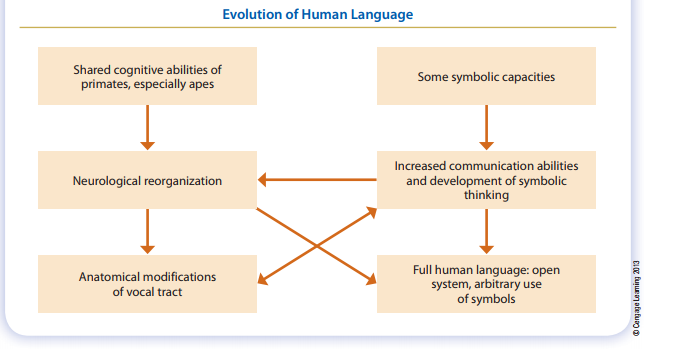
Humans use language—a set of written or spoken symbols that refer to concepts, other people, objects, and so on. This set of symbols is said to be arbitrary because the symbol itself has no inherent relationship with whatever it stands for. For example, the English word flower, when written or spoken, doesn’t look, sound, smell, or feel like the thing it represents. Humans can recombine their linguistic symbols in an infinite number of ways to create new meanings; and we can use language to refer to events, places, objects, and people far removed in both space and time. For these reasons, language is described as a form of communication based on the human ability to think symbolically.
Language, as distinct from other forms of communication, has always been considered a uniquely human achievement, setting humans apart from the rest of the animal kingdom. But work with captive apes has somewhat revised this view. Although many researchers were skeptical about the capacity of nonhuman primates to use language, reports from psychologists, especially those who work with chimpanzees, leave little doubt that apes can learn to interpret visual signs and use them in communication. Other than humans, no mammal can speak. However, the fact that apes can’t speak has less to do with lack of intelligence than to differences in the anatomy of the vocal tract and language related structures in the brain.
Beginning in the 1960s, after unsuccessful attempts to teach young chimpanzees to speak, researchers designed a study to evaluate language abilities in chimpanzees using American Sign Language for the Deaf (ASL). The research was a success, and in three years a young female named Washoe was using at least 132 signs. Years later, an infant chimpanzee named Loulis was placed in Washoe’s care. Psychologist Roger Fouts and colleagues wanted to know if Loulis would acquire signing skills from Washoe and other chimpanzees in the study group. Within just eight days, Loulis began to imitate the signs of others. Moreover, Washoe deliberately taught Loulis some signs. There have been several other chimpanzee language experiments, and work with orangutans, gorillas, and bonobos has shown that all the great apes have the capacity to use signs and symbols to communicate, not only with humans but also with each other. These abilities imply that to some degree, the great apes are capable of symbolic thought.
Questions have been raised about this type of research. Do the apes really understand the signs they learn, or are they merely imitating their trainers? Do they learn that a symbol is a name for an object or simply that using it will produce that object? Partly in an effort to address some of these questions, psychologist Sue Savage-Rumbaugh demonstrated that chimpanzees can use symbols to categorize classes of objects. Using a symbol as a label is not the same thing as understanding the representational value of the symbol; but if the chimpanzees could classify things into groups, it would indicate that they can use symbols referentially. Two chimps were taught that familiar food items (for which they used symbols) belonged to a broader category referred to by yet another symbol, “food.” Then they were introduced to unfamiliar food items, for which they had no symbols, to see if they would put them in the food category. The fact that they both had excellent scores showed While the great apes that have been involved in the language experiments have shown a remarkable degree of cognitive complexity, it nevertheless remains evident that they don’t acquire and use language in the same way humans do. It also appears that not all signing apes understand the relationship between symbol and object, person, or action. Nonetheless, there’s now abundant evidence that humans aren’t the only species capable of some degree of symbolic thought and complex communication.


The Primate Continuum
It’s an unfortunate fact that humans generally view themselves as separate from the rest of the animal kingdom. This perspective is partly due to a prevailing lack of knowledge about the behavior and abilities of other species. Moreover, these notions are continuously reinforced through exposure to advertising, movies, and television .
For decades, behavioral psychology taught that animal behavior represents nothing more than a series of conditioned responses to specific stimuli. (This perspective is very convenient for those who wish to exploit nonhuman animals, for whatever purposes,
and remain guilt-free.) Fortunately, this attitude has begun to change in recent years to reflect a growing awareness that humans, although in many ways unquestionably unique, are nevertheless part of a biological continuum and behavioral continuum. We are connected not only to our closest relatives, the other primates, but to all life on earth.
Where do humans fit in this continuum? The answer depends on the criteria used. Certainly, we’re the most intelligent species if we define intelligence in terms of problem-solving abilities and abstract thought. However, if we look more closely, we recognize that the differences between ourselves and our primate relatives, especially chimpanzees and bonobos, are primarily quantitative,
not qualitative.
Although the human brain is absolutely and relatively larger, neurological processes are functionally the same. The necessity of close bonding with at least one parent and the need for physical contact are essentially the same. Developmental stages and dependence on learning are similar. Indeed, even in the capacity for cruelty and aggression combined with compassion, tenderness, and altruism exhibited by chimpanzees, we see a close parallel to the dichotomy between “evil” and “good” so long recognized in ourselves. The main difference between how chimpanzees and humans express these qualities (and therefore the dichotomy) is one of degree. Humans are much more adept at cruelty and compassion, and we can reflect on our behavior in ways that chimpanzees can’t. Like the cat that plays with a mouse, chimpanzees don’t seem to understand the suffering they inflict on others. But humans do. Likewise, while an adult chimpanzee may sit next to a dying relative, it doesn’t seem to feel the intense grief that a human normally does in the same situation.
To arrive at any understanding of what it is to be human, it’s important to recognize that many of our behaviors are elaborate extensions of those of our hominin ancestors and close primate relatives. The fact that so many of us prefer to bask in the warmth of the “sun belt” with literally millions of others reflects our heritage as social animals adapted to life in the tropics. And the sweet tooth that afflicts so many of us is a result of our earlier primate ancestors’ predilection for high-energy sugar contained in sweet, ripe fruit. Thus, it’s important to recognize our primate heritage as we explore how humans came to be and how we continue to adapt.

Endangered Primates
In September 2000, scientists announced that a subspecies of red colobus, named Miss Waldron’s red colobus, had officially been declared extinct. This announcement came after a 6-year search for a monkey that hadn’t been seen for 20 years (Oates et al., 2000). Sadly, this monkey, indigenous to two West African countries, has the distinction of being the first nonhuman primate to be declared extinct in the twenty-first century. But it won’t be the last. In fact, as of this writing, over half of all nonhuman primate species are now in jeopardy, and some face almost certain extinction in the wild.
There are three basic reasons for the worldwide depletion of nonhuman primates: habitat destruction, human hunting, and live capture for export or local trade. Underlying these three causes is one major factor, unprecedented human population growth, particularly in developing countries, where most nonhuman primates live.
The developing nations of Africa, Asia, and Central and South America are home to over 90 percent of all nonhuman primate species. During the 1990s, these countries, aided by Europe, China, and the United States, destroyed an average of 39 million acres of forest per year. The destruction declined between 2000 and 2010 to about 32 million acres a year, largely because of restrictions in Brazil. (Food and Agriculture Organization of the United Nations, 2010). But whether these restrictions will hold remains to be seen. Tropical forests are cleared for agriculture, pasture, lumber, and large-scale mining operations (with their necessary roads). Moreover, millions of people in many developing countries are critically short of fuel; lacking electricity, they have a critical need for firewood. Lastly, the demand for tropical hardwoods (such as mahogany, teak, and rosewood) in the United States, Europe, and Japan continues unabated, creating an enormously profitable market for rain forest products. The need for wood has resulted in a conflict with conservationists, especially in central Africa. Mountain gorillas are one of the most endangered nonhuman primate species, and tourism has been the only real hope of salvation for these magnificent animals. For this reason, several gorilla groups have been habituated to humans and are protected by park rangers. Nevertheless, poaching, civil war, and land clearing have continued to take a toll on these small populations. For example, between January and late July 2007, 10 mountain gorillas were shot in the Virunga Volcanoes Conservation Area (shared by Uganda, Rwanda, and the DRC; Fig. 6-38). The gorillas weren’t shot for meat or because they were raiding crops. They were shot because the presence and protection of mountain gorillas are obstacles to people who would destroy what remains of the forests. But gorillas are not the only primates to have been shot in the Virungas. In the past few years, more than 120 rangers have been killed while protecting wildlife.
Habitat loss used to be the single greatest threat to nonhuman primates. But in the past few years, human hunting has perhaps become an even more important factor. During the 1990s, primatologists and conservationists became aware of a rapidly developing trade in bushmeat, meat from wild animals, especially in Africa. The current slaughter, which now accounts for the loss of tens of thousands of nonhuman primates and other animals annually, has been compared to the near extinction of the American bison in the nineteenth century. Wherever primates live, people have always hunted them for food. But in the past, subsistence hunting wasn’t a serious threat to nonhuman primate populations, and certainly not to entire species.
But now, hunters armed with automatic rifles can, and do, wipe out an entire group of monkeys or gorillas in minutes. In fact, it’s now possible to buy bushmeat in major cities throughout Europe and the United States. Illegal bushmeat is readily available to immigrants who want traditional foods and to nonimmigrants who think that it’s trendy to eat meat from exotic, and frequently endangered, animals. It’s impossible to know how many animals are killed each year, but the estimates are staggering. The Society for Conservation Biology estimates that about 6,000 kg (13,228 pounds) of bushmeat are taken through just seven western cities (New York, London, Toronto, Paris, Montreal, Chicago, and Brussels) every month. No one knows how much of this meat is from primates, but this figure represents only a tiny fraction of all the animals being slaughtered because much smuggled meat isn’t detected. Also, the international trade is thought to account for only about 1 percent of the total (Marris, 2006). It’s difficult to comprehend, but within a relatively short period of time, hunting wild animals for food, particularly in Africa, has shifted from being a subsistence activity to a commercial enterprise of international scope.
Although the slaughter may be best known in Africa, it’s by no means limited to that continent. Hunting and live capture of endangered primates continues unabated in China and Southeast Asia, where nonhuman primates are not only eaten but also funneled into the exotic pet trade. But just as important, primate body parts figure prominently in traditional medicines. With increasing human population size, the enormous demand for these products (and products from other, nonprimate species, such as tigers) has put many species in extreme jeopardy. With some species numbering only a few hundred or a few thousand animals, nonhuman primates cannot survive this onslaught for more than a few years.
Primates are also captured live for zoos, biomedical research, and the exotic pet trade As a result of their mothers being captured,
hundreds of infants are orphaned and end up sold in markets as pets. Although a few of these traumatized orphans make it to sanctuaries, most die within days or weeks of capture . Live capture has declined since the Convention on International Trade in Endangered Species of Wild Flora and Fauna (CITES) was implemented in 1973. By August 2005, a total of 169 countries had signed this treaty, agreeing not to allow trade in species listed by CITES as being endangered . However, even some CITES members are still occasionally involved in the illegal primate trade (Japan and Belgium, among others). As a note of optimism, in November 2007, the DRC government and the Bonobo Conservation Initiative (in Washington, D.C.) created a bonobo reserve consisting of 30,500 km2. This amounts to about 10 percent of the land in the DRC, and the government has stated that its goal is to set aside an additional 5 percent for wildlife protection (News in Brief, 2007). This is a huge step forward, but it remains to be seen if protection can be enforced. There are many conservation groups working to protect nonhuman primates. These include, among many others, Conservation International, the World Wildlife Fund, and the Jane Goodall Institute. It goes without saying that these and other organizations must succeed if the great apes are to survive in the wild even until the middle of this century. If you are in your 20s or 30s, you will certainly live to hear of the extinction of some of our marvelous cousins. Many more will undoubtedly slip away unnoticed. Tragically, this will occur, in most cases, before we’ve even gotten to know them. Each species on earth is the current result of a unique set of evolutionary events that, over millions of years, has produced a finely adapted component of a diverse, interconnected ecosystem. When it becomes extinct, that adaptation and that part of biodiversity is lost forever. What a tragedy it will be if, through our own mismanagement and greed, we awaken to a world without chimpanzees, mountain gorillas, or the tiny, exquisite lion tamarin. When that day comes, we truly will have lost a part of ourselves, and we will certainly be the poorer for it.