Mutation: When Genes Change
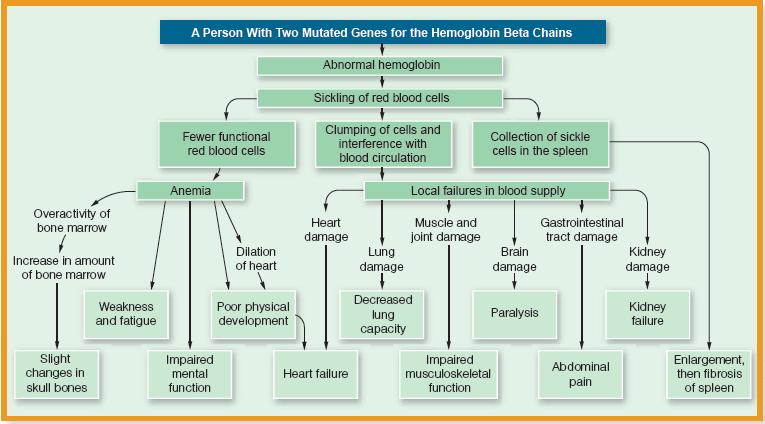
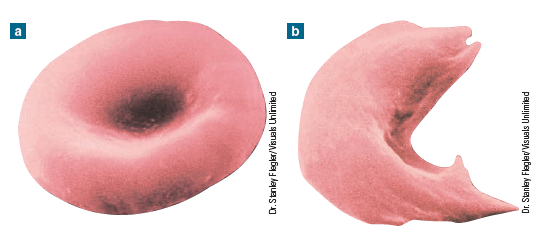
The best way to understand how genes function is to see what happens when they change, or mutate. Normal adult haemoglobin is made up of four amino acid chains (two alpha chains and two beta chains) that are the direct products of gene action. Each beta chain is in turn composed of 146 amino acids. There are several hemoglobin disorders with genetic origins, and perhaps the best known of these is sickle-cell anemia, which results from a defect in the beta chain. People with sickle-cell anemia inherit, from both parents, a mutated form of the gene that directs the formation of the beta chain. This mutation is caused by the substitution of one amino acid (value) for the amino acid that’s normally present (glutamic acid). This single amino acid substitution on the beta chain results in the production of a less efficient form of hemoglobin called hemoglobin S (HbS) instead of the normal form, which is called hemoglobin A (HbA). In situations where the availability of oxygen is reduced, such as at high altitude or when oxygen requirements are increased through exercise, red blood cells with HbS collapse and become sickle-shaped . What follows is a cascade of events, all of which result in severe anemia and its consequences . Briefly, these consequences include impaired circulation from blocked capillaries, red blood cell destruction, oxygen deprivation to vital organs (including the brain), and, without treatment, death. People who inherit the altered form of the gene from only one parent don’t have sickle-cell anemia, but they do have what’s called sickle-cell trait. Fortunately for them, they’re much less severely affected because only about 40 percent of their hemoglobin is abnormal.
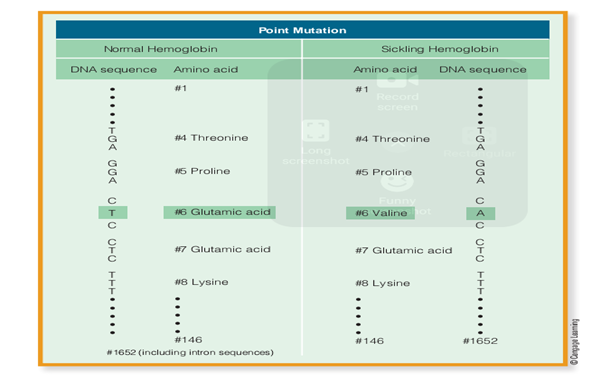
The cause of sickle-cell anemia is a very slight change in the Hb gene. Remember that hemoglobin beta chains each have 146 amino acids. What’s more, to emphasize the importance of a seemingly minor alteration, consider that triplets of DNA bases are required to specify amino acids. Therefore it takes 438 bases (146 × 3) to produce the chain of 146 amino acids that forms the adult haemoglobin beta chain. But a change in only one of these 438 bases produces the life threatening amino acid products for both normal and sickling hemoglobin. As you can see, a single base substitution (from CTC to CAC) can result in an altered complications seen in sicklecell anemia. Below figure shows the DNA base sequence and the resulting amino acid sequence, from proline—glutamic acid—glutamic acid . to proline—valine—glutamic acid . . . This kind of change in the genetic code is referred to as a point mutation or base substitution. In evolution, these changes are important sources of new genetic variation in populations. Point mutations, like the one that causes sickle-cell anemia, probably occur fairly frequently. But for a new mutation to be evolutionarily significant, it must be passed on to offspring and eventually become more common in a population.
Once point mutations occur, their fate in populations depends on the other evolutionary forces, especially natural selection. Depending on how beneficial a mutation is, it may become more common over time; if it’s disadvantageous, it probably won’t. Sickle-cell anemia is one of the best examples of natural selection acting on humans; it shows us how a disadvantageous mutation can become more frequent in certain environments.