Infection, as opposed to other disease categories such as degenerative or genetic disease, includes pathological conditions caused by microorganisms (viruses, bacteria, fungi, and other one-celled organisms). Throughout the course of human evolution, infectious disease has exerted enormous selective pressures on populations, influencing the frequency of alleles that affect the immune response. Indeed the importance of infectious disease as an agent of natural selection in human populations cannot be overemphasized. But as important as infectious disease has been, its role in this regard isn’t very well documented.
The effects of infectious disease on humans are mediated culturally as well as biologically. Innumerable cultural factors, such as architectural styles, subsistence techniques, exposure to domesticated animals, transportation, and even religious practices, affect how infectious disease develops and persists within and between populations.
Until about 15,000 years ago, all humans lived in small nomadic hunting and gathering groups. These groups rarely stayed in one location more than a few days or weeks at a time, so they had little contact with refuse heaps that house disease vectors. But with the domestication of plants and animals, people became more sedentary and began living in small villages. Gradually, villages became towns; and towns, in turn, developed into densely crowded, unsanitary cities.
As long as humans lived in small bands, there was little opportunity for infectious disease to affect large numbers of people. Certainly people were sometimes infected with various pathogens through contact with animals they killed and butchered; they were also exposed to infectious illness by drinking contaminated water, exposure to insects such as mosquitoes, and simply through association with each other. But even if an entire local group or band were wiped out, the effect on the overall population in a given area would have been negligible. Moreover, for a disease to become endemic in a population, there must be enough people to sustain it. Therefore small bands of hunter-gatherers weren’t faced with continuous exposure to endemic disease.
But with the advent of settled living and close proximity to domesticated animals, opportunities for exposure to disease increased. As sedentary life permitted larger group size, it became possible for several diseases to become permanently established in some populations. Moreover, exposure to domestic animals, such as cattle and fowl, provided an opportune environment for the spread of several zoonotic diseases. The crowded, unsanitary conditions that characterized parts of all cities until the late nineteenth century and that still persist in much of the world today further added to the disease burden borne by human populations.
Tuberculosis, discussed above, has been one of the most cited examples of zoonotic disease. It is believed to have been transmitted from cattle to humans after cattle were domesticated some 10,000 ya. In fact, TB is widely considered to be one of the many prices humans have paid for living in close association with domesticated animals. Mycobacterium tuberculosis usually infects the lungs. It’s spread through sneezing and coughing, and symptoms include coughing, fatigue, and fever. Prior to the development of antibiotic therapies, the disease often proved fatal. In addition to the lungs, the bacterium can attack other tissues, including bone. The area of the skeleton most commonly involved is the spine and when this occurs, two or three vertebrae may be destroyed and eventually collapse (Fig. 15-8).
Malaria provides perhaps the bestdocumented example of how disease can act to change allele frequencies in human populations. In Chapter 4, you saw how, in some African and Mediterranean populations, malaria has altered allele frequencies at the locus that governs hemoglobin formation, leading to an increased prevalence of sickle-cell anemia. Despite extensive long-term eradication programs, malaria still poses a serious threat to human health. Indeed, the World Health Organization estimates the number of people currently infected with malaria to be between 300 and 500 million worldwide. And this number is increasing as drug-resistant strains of the disease-causing microorganism become more common (Olliaro et al., 1995).
Another example of the selective role of infectious disease is indirectly provided by AIDS (acquired immunodeficiency syndrome). In the United States, the first cases of AIDS were reported in 1981. Since then, perhaps as many as 1.5 million Americans have been infected by HIV (human immunodeficiency virus), the agent that causes AIDS. However, most of the burden of AIDS is borne by developing countries, where 95 percent of all HIV-infected people live (Fig. 15-9). According to World Health Organization estimates, between 32 and 35 million people worldwide were living with HIV infection as of November 2010, and more than 25 million had died (UNAIDS/WHO 2010 AIDS Epidemic Updates).
By the early 1990s, scientists were aware of some patients who had been HIV-positive for 10 to 15 years but continued to show few if any symptoms, leading researchers to suspect that some individuals were naturally resistant to HIV. This was shown to be true in late 1996 with the publication of two independent studies that demonstrated a mechanism for HIV resistance (Dean et al., 1996; Samson et al., 1996).
These two reports describe a genetic mutation that involves a major receptor site on the surface of certain immune cells, including T4 cells. (Receptor sites are protein molecules that enable HIV and other viruses to invade cells.) As a result of the mutation, the receptor site doesn’t function properly and the virus can’t enter the cell. Current evidence strongly suggests that people who are homozygous for this allele may be completely resistant to many types of HIV infection. In heterozygotes, infection may still occur, but the course of HIV disease is significantly slowed.
For unknown reasons, the mutant allele occurs mainly in people of European descent, among whom its frequency is about 10 percent. However, the mutation appears to be absent in certain Japanese and West African groups that were studied (Samson et al., 1996). Another research team reported an allele frequency of about 2 percent among African Americans; they speculated that the presence of the allele in African Americans is due to genetic admixture (gene flow) with European Americans (Dean et al., 1996). They also suggested that this polymorphism exists in Europeans because of selective pressures favoring an allele that originally occurred as a rare mutation. But it’s important to understand that the original selective agent was not HIV. Instead, it was some other as yet unidentified pathogen that requires the same receptor site as HIV, and some researchers have implicated the virus that causes smallpox. Lalani and colleagues (1999) reported that a poxvirus. related to the virus that causes smallpox, can use the same receptor site as HIV. While this conclusion hasn’t yet been firmly established, it offers a very interesting avenue of research. It may reveal how a mutation that has been favored by selection because it provides protection against one type of infection (perhaps smallpox) can also increase resistance to another (AIDS).
Smallpox, once a deadly viral disease, is estimated to have accounted for 10 to 15 percent of all deaths in parts of Europe during the eighteenth century (Fig. 15-10). It’s possible that during its long history, smallpox may have altered the frequency of the ABO blood types by selecting against the A allele. Smallpox had a higher incidence in people with blood type A or AB than in type O individuals, a fact that may be explained by the presence of an antigen on the smallpox virus that’s similar tothe A antigen. Thus, when some type A individuals were exposed to smallpox, their immune systems failed to recognize the virus as foreign and didn’t mount an adequate immune response. This meant that people with the A allele died in greater numbers than those without it. So in regions where smallpox was common in the past, it could have altered allele frequencies at the ABO locus by selecting against the A allele.
Smallpox, once a devastating killer of millions, is the only condition to have been successfully eliminated by modern medical technology. By 1977, through massive vaccination programs, the World Health Organization was able to declare the smallpox virus extinct except for a few colonies in research labs in the United States and Russia.
It’s important to understand that humans and pathogens exert selective pressures on each other, creating a dynamic relationship between disease organisms and their human (and nonhuman) hosts. Just as disease exerts selective pressures on host populations to adapt, microorganisms also evolve and adapt to various pressures exerted on them by their hosts.
Evolutionarily speaking, it’s to the advantage of any pathogen not to be so deadly that it kills its host too quickly.If the host dies shortly after becoming infected, the virus or bacterium may not have time to reproduce and infect other hosts. Thus selection sometimes acts to produce resistance in host populations and/or to reduce the virulence of disease organisms, to the benefit of both. However, members of populations exposed for the first time to a new disease frequently die in huge numbers. This type of exposure was a major factor in the decimation of indigenous New World populations after Europeans introduced smallpox into Native American groups. And it has also been the case with the current worldwide spread of HIV.Of the known disease-causing organisms, HIV provides the best- documented example of evolution and adaptation in a pathogen. It’s also one of several examples of interspecies transfer of infection. For these reasons, we focus much of this discussion of evolutionary factors and infectious disease on HIV.
The type of HIV responsible for the AIDS epidemic is HIV-1, which is extremely variable genetically. Since the late 1980s, researchers have been comparing the DNA sequences of HIV and a closely related virus called simian immunodeficiency virus (SIV), which is found in chimpanzees and several African monkey species. Like HIV, SIV is genetically variable, and each strain appears to be specific to a given primate species. SIV produces no symptoms in the African monkeys and chimpanzees that are its traditional hosts, but when injected into Asian monkeys, it eventually causes immune suppression, AIDS-like symptoms, and death. These findings indicate that the various forms of SIV have shared a long evolutionary history with a number of African primate species and that these primates have developed ways of accommodating this virus, which is deadly to their Asian relatives. These results also substantiate long-held hypotheses that SIV and HIV evolved in Africa. Furthermore, DNA comparisons have shown that HIV-1 almost certainly evolved from the form Of SIV that infects chimpanzees indigenous to western central Africa (Gao et al., 1999).
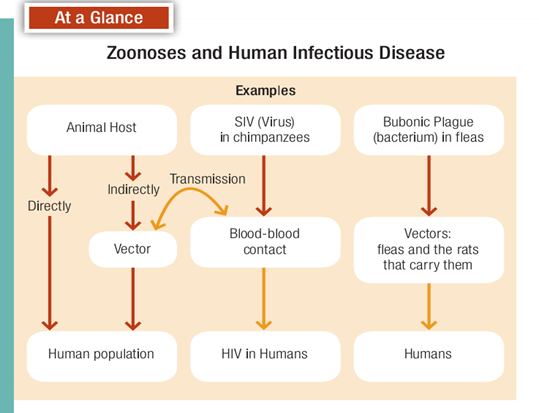
As you read earlier, in parts of West Africa, chimpanzees are routinely hunted by humans for food. So the most probable explanation for the transmission of SIV from chimpanzees to humans is the hunting and butchering of chimpanzees (Gao et al., 1999; Weiss and Wrangham, 1999). Thus, HIV/AIDS is a zoonotic disease (Fig. 15-11). The DNA evidence further suggests that there were at least three separate human exposures to chimpanzee SIV, and at some point the virus was altered to the form we call HIV. Exactly when chimpanzee SIV was transmitted to humans is unknown. The oldest evidence of human infection is a frozen HIV-positive blood sample taken from a West African patient in 1959. Therefore, although human exposure to SIV/HIV probably occurred many times in the past, the virus didn’t become firmly established in humans until the latter half of the twentieth century.
Influenza is a contagious respiratory disease caused by various strains of virus. It, too, is a zoonotic disease, and it has probably killed more humans than any other infectious disease. There were two flupandemics in the twentieth century; the first of these killed an estimated 20 million people in 1918. Moreover, “seasonal flu,” which comes around every year, killed approximately 36,000 people annually in the United States during the 1990s (Centers for Disease Control, 2009). Worldwide, it accounts for several hundred thousand deaths every year.
The influenza viruses that infect humans are initially acquired through contact with domestic pigs and fowl (Fig. 15-12). For this reason, influenza is frequently referred to as swine or avian (bird) flu, depending on which species transmitted it to humans. In 2009, a new swine flu virus called H1N1 caused great fear of another pandemic, partly because it caused more severe illness in younger people than most flu viruses.
Because swine flu epidemics are less frequent than the seasonal avian flu, people have less resistance when confronted with a “new” swine flu virus. Swine flu can also be more deadly, and health professionals are always mindful of, and haunted by, the memory of the catastrophic 1918 pandemic. For all these reasons, health professionals worldwide mobilized an enormous effort in 2009 to prepare for a new swine flu pandemic. Hundreds of millions of doses of vaccine were distributed, but the epidemic proved not to be as severe as originally feared. Still, health officials are always on the alert for the possibility of an influenza pandemic, partly because of the ever-present danger posed by close contact between humans, pigs, and domestic fowl.
Until the twentieth century, infectious disease was the number one cause of death in all human populations. Even today in many developing countries, as much as half of all mortality is due to infectious disease, compared with only about 10 percent in the United States. For example, there are an estimated 1 million deaths due to malaria every year. That figure computes to one malaria-related death every 30 seconds (Weiss, 2002)! Ninety percent of these deaths occur in sub-Saharan Africa, where 5 percent of children die of malaria before age 5 (Greenwood and Mutabingwa, 2002; Weiss, 2002). In the United States and other industrialized nations, with improved living conditions, better sanitation, and the widespread use of antibiotics since the 1940s, infectious disease has given way to heart disease and cancer as the leading causes of death.
Optimistic predictions held that infectious disease would one day be a thing of the past. You may be surprised to learn that in the United States mortality due to infectious disease has actually increased in recent years (Pinner et al., 1996). This increase may partly be due to the overuse of antibiotics. It’s estimated that half of all antibiotics prescribed in the United States are used to treat viral conditions such as colds and flu. Because antibiotics are completely ineffective against viruses, antibiotic therapy in these cases is not only useless but may actually have dangerous long-term consequences. There’s considerable concern in the biomedical community over the indiscriminate use of antibiotics since the 1950s. Antibiotics have exerted selective pressures on bacterial species that have, over time, evolved antibioticresistant strains (an excellent example of natural selection). So in the past few years, we’ve seen the reemergence of many bacterial diseases, including, pneumonia, cholera, and TB in forms that are less responsive to treatment.
The World Health Organization now lists tuberculosis as the world’s leading killer of adults (Colwell, 1996). In fact, the number of TB cases worldwide has risen 28 percent since the mid-1980s, with an estimated 10 million people infected in the United States alone. Although not all infected people develop active disease, in the 1990s an estimated 30 million persons worldwide are believed to have died from TB. One very troubling aspect of the increase in TB infection is that new strains of Mycobacterium tuberculosis are resistant to many antibiotics and other treatments.
Various treatments for nonbacterial conditions have also become ineffective. One such example is the appearance of chloroquine-resistant malaria, which has rendered chloroquine (the traditional preventive medication) virtually useless in some parts of Africa. And many insect species have also developed resistance to commonly used pesticides.
Fundamental to all these factors is human population growth. As it continues to soar, it causes more environmental disturbance and, through additional human activity, increased global warming. Global warming is important in this regard because, as temperatures rise in previously cooler regions and as patterns of precipitation change, new habitats for disease vectors such as mosquitoes open up. Moreover, in developing countries, where as much as 50 percent of mortality is due to infectious disease, overcrowding and unsanitary conditions increasingly contribute to higher rates of communicable illness. It’s hard to conceive of a better set of circumstances for the appearance and spread of communicable disease, and it remains to be seen if scientific innovation, medical technology, and political views will be able to meet the challenge.