Since the discovery of DNA structure and function in the 1950s, the field of genetics has revolutionized biological science and reshaped our understanding of inheritance, genetic disease, and evolutionary processes. For example, a technique developed in 1986, called polymer chain reaction (PCR), enables scientists to make thousands of copies of small samples of DNA, which can then be analysed. In the past, DNA samples from crime scenes or fossils were usually too small to be studied. But PCR makes it possible to examine DNA sequences in, for example, Neanderthal fossils and Egyptian mummies, and it has limitless potential for many disciplines, including forensic science, medicine, and palaeoanthropology.
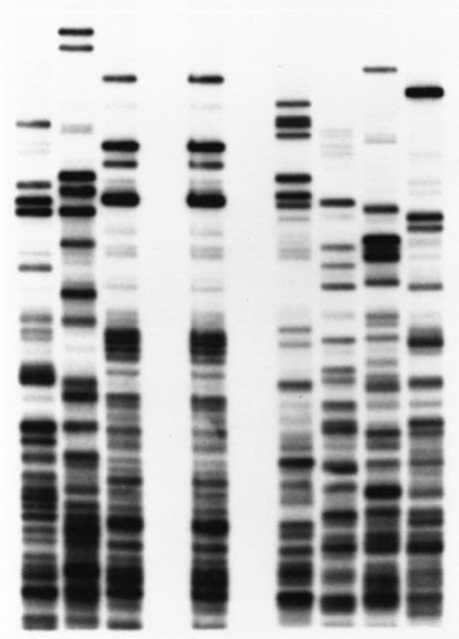
Another application of PCR allows scientists to identify DNA fingerprints, so called because they appear as patterns of repeated DNA sequences that are unique to each individual (Fig. 3-22on p. 73 ). For example, one person might have a segment of six bases such as ATTCTA repeated 3 times, while another person might have 20 copies of the same sequence. DNA fingerprinting is perhaps the most powerful tool available for human identification. Scientists have used it to identify scores of remains, including members of the Russian royal family murdered in 1918 and victims of the September 11, 2001, terrorist attacks. Moreover, the technique has been used to exonerate many innocent people wrongly convicted of crimes, in some cases decades after they were imprisoned.
Over the last two decades, scientists have used the techniques of recombinant DNA technology to transfer genes from the cells of one species into those of another. One common method has been to insert human genes that direct the production of various proteins into bacterial cells in laboratories. The altered bacteria can then produce human gene products, such as insulin. Until the early 1980s, people with diabetes relied on insulin derived from nonhuman animals. However, this insulin wasn’t plentiful, and some patients became allergic to it. But since 1982, abundant supplies of human insulin, produced by bacteria, have been available, and insulin derived from bacteria doesn’t cause allergic reactions.
In recent years, genetic manipulation has become increasingly controversial owing to questions related to product safety, environmental concerns, animal welfare, and concern over the experimental use of human embryos. For example, the insertion of bacterial DNA into certain crops has made them toxic to leaf-eating insects, thus reducing the need for pesticides. Cattle and pigs are commonly treated with antibiotics and genetically engineered growth hormone to increase growth rates. (There’s no concrete evidence that humans are susceptible to the insect-repelling bacterial DNA or harmed by consuming meat and dairy products from animals treated with growth hormone. But there are concerns over the unknown effects of long-term exposure.)
Cloning has been one of the most controversial of all the new genetic technologies. But cloning isn’t as new as you might think. Anyone who has ever taken a cutting from a plant and rooted it to grow a new one has produced a Table clone. Many mammalian species have now been cloned, and researchers have even produced clones of dead mice that were frozen for as long as 16 years. This gives rise to hopes that eventually it may be possible to clone extinct animals, such as mammoths, from the frozen bodies of animals that died several thousand years ago (Wakayama et al., 2008). But don’t count on visiting a Jurassic Park type zoo anytime soon. As exciting as these innovations are, probably the single most important advance in genetics has been the progress made by the Human Genome Project (International Human Genome Sequencing Consortium, 2001; Venter et al., 2001). The goal of this international effort, begun in 1990, was to sequence the entire human genome, which consists of some 3 billion bases making up approximately 21,000 protein-coding genes. This extremely important project was completed in 2003. Since that time, the genomes of hundreds of species have been sequenced, including those of chimpanzees (Chimpanzee Sequencing and Analysis Consortium, 2005), rhesus macaques (Rhesus Macaque Genome Sequencing and Analysis Consortium, 2007), western lowland gorillas (Scally et al., 2012), Orangutans (Locke et al., 2011), and bonobos (Prüfer et al., 2012). By comparing different primate genomes, including that of humans, molecular anthropologists are revealing more details regarding phylogenetic relationships among all primate species.
Since the publication of the human genome, DNA sequencing technologies have become increasingly inexpensive, more widely available, and much faster. In May 2010, researchers finished sequencing the entire Neandertal genome (Green et al., 2010). To date, the most exciting announcement stemming from this research is that modern Europeans and Asians (but not Africans) inherited 1 to 4 percent of their genes from ancient Neandertal ancestors. This finding sheds light on debates concerning whether or not early modern humans interbred with Neandertals. These debates have been ongoing in physical anthropology for more than 50 years, and while this new genetic evidence does not conclusively end the discussion, it strongly supports the argument that interbreeding did indeed take place and that many of us carry a few Neandertal genes . Equally exciting was the sequencing of the entire genome of another pre-modern human group called the Denisovans (Reich et al., 2010; Meyer et al., 2012). This group is dated to around 50,000 ya and the entire col-lection of Denisovan skeletal remains consists of a tiny finger bone and two teeth discovered in Siberia. Yet, in a feat that would have been unimaginable just ten years ago, researchers have been able to obtain high quality DNA from the finger bone and sequence the entire genome of this population! The Denisovan genome has now been compared to that of their Neandertal cousins and to the genomes of modern human populations .
Eventually, comparative genome analysis should provide a thorough assessment of genetic similarities and differences and thus of the evolutionary relationships between humans and other primates. What’s more, we can already look at human variation in an entirely different light than was possible just 10 years ago (see Chapter 15). Among other things, genetic comparisons between human groups can inform us about population movements in the past and what selective pressures may have been exerted on different populations to produce some of the variability we see.
The Encyclopaedia of DNA Elements, or ENCODE, is a project initially conceived to follow up on the progress made by the Human Genome Project. This huge study, begun in 2003, now involves an international consortium of more than 400 researchers who, in September 2012, simultaneously published 30 articles in several scientific journals. Initially, the project set out to catalog the functional DNA sequences contained within the vast stretches of non-protein coding DNA, determine what they do, and examine how the human genome is regulated (Maher, 2012; Pennisi, 2012; The ENCODE Project Consortium, 2012). The current results of the ENCODE project are far too numerous to mention here. But the some of the most important ones include the discovery that as much as 80 percent of the human genome is involved in some form of biochemical function. That estimate may be high and most of the biochemical functions have not been identified. In fact, some of them are probably not even important. But some of these activities include the manufacture of non-coding RNA (RNA that is not involved in protein synthesis but that regulates gene function), and binding sites where regulatory proteins attach, In short, some of what used to be called “junk DNA” is active in gene regulation. It is also of great interest that many regulatory factors have been shown to be involved in disease, including autoimmune conditions such as rheumatoid arthritis, Crohn’s disease, and multiple sclerosis (Maurano, et al., 2012). This discovery will radically alter future approaches to the diagnosis and treatment of genetically caused diseases
The ENCODE project has determined that at least 9 percent (and probably much more) of the human genome has regulatory functions. It has also determined that the regulation of protein coding genes is more complex and has evolved more quickly in humans than in most other species. Moreover, evolution occurs more rapidly in regulatory elements. These facts may partly explain the accelerated pace of evolutionary change in modern humans compared to that of other animals. Ultimately the detailed understanding of how gene regulation works will revolutionize how we view evolutionary processes. The focus of many genetic and evolutionary studies will shift away from protein coding genes and toward regulatory elements. The factors that regulate gene activity in embryonic development are also the basis of evolutionary change. It follows that if we are going to reveal the secrets of our evolutionary past, we must first examine how DNA function is regulated in the present. Certainly the term “junk DNA” will be laid to reset